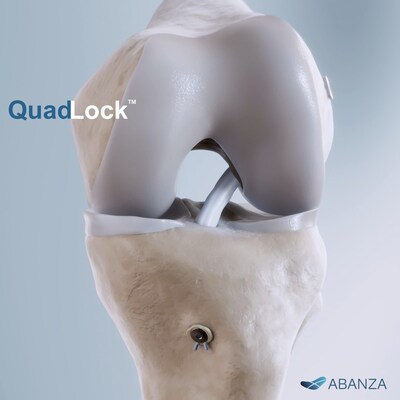
ORLANDO, Fla., Jan. 27, 2026 /PRNewswire/ — ABANZA, a company focused on next-generation soft-tissue repair solutions, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for QuadLock™, its new knotless, bidirectional tension-adjustable fixation system for ACL reconstruction.
QuadLock™ is designed to securely fixate sutures and tapes, helping surgeons fine-tune graft tension and maintain stability across several graft configurations commonly used in ACL procedures, including quadriceps tendon, quadrupled semitendinosus/gracilis, and bone–patellar tendon–bone (BTB).
In biomechanical testing under high-demand cyclic loading, QuadLock™ demonstrated market-leading control of cyclic displacement (less than 0.5 mm)—a 500%+ improvement compared to the 3–6 mm reported for conventional fixation methods such as cortical buttons and interference screws. QuadLock™ also achieved a pullout strength of >1,000 N, combining high fixation strength with minimal displacement. Avoiding loss of tension under repeated loading is critical in early recovery, when fixation stability directly influences the restoration of functional joint stability.
ACL reconstruction remains a major procedure in sports medicine, with approximately 300,000 surgeries performed each year in the United States. With its combination of minimal cyclic displacement, knotless fixation, and tension adjustability, QuadLock™ expands surgeons’ options for consistent graft management across a broad range of patient bone quality.
“QuadLock™ reflects our focus on practical innovation—giving surgeons adjustability, control, and confidence when fixation performance matters most, especially as today’s patient population continues to evolve and traditional fixations are increasingly challenged,” said Juan Abascal, Chief Executive Officer of ABANZA. “This clearance strengthens our U.S. roadmap and supports our broader platform strategy to advance soft-tissue repair with solutions that emphasize reproducible technique and stable fixation under demanding conditions.”
QuadLock™ is part of ABANZA’s growing platform of soft-tissue fixation technologies. Additional products in development—including LoopCap™ and WasherCap™ In Line— are intended to extend the portfolio into indications such as biceps tendon repair and AC Joint reconstruction, MPFL and LET repair, and select Foot and Ankle applications including Syndesmosis and Achilles repair.
For more information about QuadLock™ and ABANZA’s product pipeline, visit www.abanzamed.com.
About ABANZA
ABANZA is advancing soft-tissue repair with a new generation of fixation technologies designed to help surgeons deliver reliable outcomes for today’s evolving patient population. Our devices are engineered to support modern surgical techniques and help surgeons achieve consistent, reliable results in soft-tissue repair.
Driven by a commitment to clinical excellence and continuous innovation, ABANZA is building a platform of solutions that aims to raise the standard of care and expand what’s possible in soft-tissue repair.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/abanza-secures-fda-510k-clearance-for-quadlock-fixation-system-for-acl-reconstruction-302669205.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/abanza-secures-fda-510k-clearance-for-quadlock-fixation-system-for-acl-reconstruction-302669205.html
SOURCE Abanza, Inc.