- Pivotal Phase II/III clinical trial underway, aiming for US and European market approval in 2028
- A ‘truly transformative’ regenerative medicine targeting a multi-million patient population with no existing curative treatment
- Fully biological transplant eliminates need for immunosuppression and restores natural function
- Scalable process protected by 92 granted patents
- CVI market projected to reach USD 5,951 million by 2034*
- Supported by USD 10 million financing from existing investors
GOTHENBURG, Sweden, March 25, 2026 /PRNewswire/ — VERIGRAFT, a Swedish clinical-stage biotechnology company pioneering personalized, fully biological therapies, today announced the initiation of a pivotal Phase II/III trial of its P-TEV (personalized tissue engineered vein) in patients with chronic venous insufficiency (CVI), supported by USD 10 million from existing investors.
The milestone comes at a time when regenerative medicine and personalized biologics are gaining significant traction among global investors and healthcare systems.
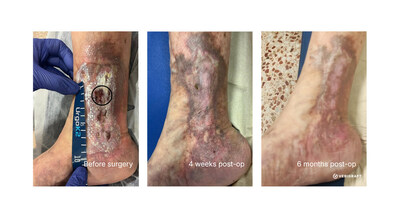
VERIGRAFT’s fully biological approach opens the way for a potential curative treatment for CVI, using personalized tissues that eliminates the need for immunosuppressive therapy. The manufacturing process is completed within 10 days, making VERIGRAFT’s offering both scalable and clinically feasible. Trial sites for the Phase II/III study are located in Spain, the Netherlands and Poland.
As an advanced therapy medicinal product (ATMP), VERIGRAFT’s clinical development programme is designed to generate robust clinical data in a limited patient population. The Company aims to achieve market approval in Europe and the US in 2028.
Chronic Venous Insufficiency (CVI) is a progressive and under-recognized vascular disease in which the veins in the legs fail to efficiently return blood to the heart, leading to sustained elevated venous pressure in the leg veins. Driven by factors such as ageing, obesity, and sedentary lifestyles, CVI represents a growing healthcare burden worldwide.
VERIGRAFT’s personalized tissue-engineered vein (P-TEV) is designed to replace diseased venous segments with dysfunctional valves with vein segments containing functioning valves. The approach uses donor-derived venous material that is decellularized and subsequently personalized using the patient’s own blood, creating a patient-specific graft. Once prepared, the P-TEV is surgically implanted to replace the diseased vein segment and restore venous function.
Petter Björquist, CEO of VERIGRAFT, said: “This financing enables a critical step forward in our clinical development and is a critical component in our journey to redefine how vascular disease can be treated by restoring natural biological function at scale. With P-TEV, we are addressing a large, underserved patient population with a therapy designed to address the underlying cause of disease, not just manage the symptoms. The initiation of our pivotal trial brings us significantly closer to making this a clinical reality.”
Dr. Bryan Kluck, an Interventional Cardiologist, Endovascular Interventionalist and Vascular Medicine Specialist at Lehigh Valley Hospital, Allentown, PA, commented: “As an interventional cardiologist familiar with the technology, I believe it offers a new way forward. This is truly restorative, unlike so many of the available tools. Patency of the venous system, along with functional vein valves, has the potential to revolutionize venous therapy. This technology restores normal venous hemodynamics, a concept that, heretofore has been unimaginable.”
Dr. Andrés García León, Head of the Department of Angiology and Vascular Surgery at Virgen de Valme University Hospital, Associate Professor of the University of Seville, and Lead Principal Investigator (PI), added: “I am very pleased to be leading the phase II/III trial of P-TEV. In clinical practice, treatment options for patients with advanced CVI are limited. P-TEV represents a promising and scalable approach with the potential to restore physiological blood flow.”
*Chronic Venous Insufficiency Market Insight, Epidemiology And Market Forecast – 2034
Source: DelveInsight
Visit www.verigraft.com
Photo: https://mma.prnewswire.com/media/2942028/VERIGRAFT_P_TEV.jpg
Logo: https://mma.prnewswire.com/media/2942027/VERIGRAFT_Logo.jpg
![]() View original content to download multimedia:https://www.prnewswire.co.uk/news-releases/verigraft-advances-first-potential-curative-treatment-for-cvi-into-pivotal-phase-iiiii-trial-302724648.html
View original content to download multimedia:https://www.prnewswire.co.uk/news-releases/verigraft-advances-first-potential-curative-treatment-for-cvi-into-pivotal-phase-iiiii-trial-302724648.html